Modern eCOA Solution for Faster Studies
With our eCOA solution at the core, sponsors and CROs rely on uMotif’s scalable and configurable platform to deliver critical data capture.


Other electronic clinical outcome assessments simply digitize paper forms; uMotif’s eCOA solutions are designed for patient centricity and more efficient, higher-quality trials. Our highly configurable, scalable platform combines intuitive user experience with behavioral science and seamlessly integrates ePRO, wearables and real-world data to reduce site and patient burden. We offer a modern, unified experience with >90% compliance rates in both clinical trials and real-world studies, delivering richer, more complete datasets faster.
Capture all types of eCOA data, on any device
uMotif’s modern platform supports both app and web-based data capture using provisioned devices or bring your own device (BYOD), to quickly build and deploy any type of eCOA to support all your study needs.
- ePRO
- eClinRO
- eObsRO
- ePerfO

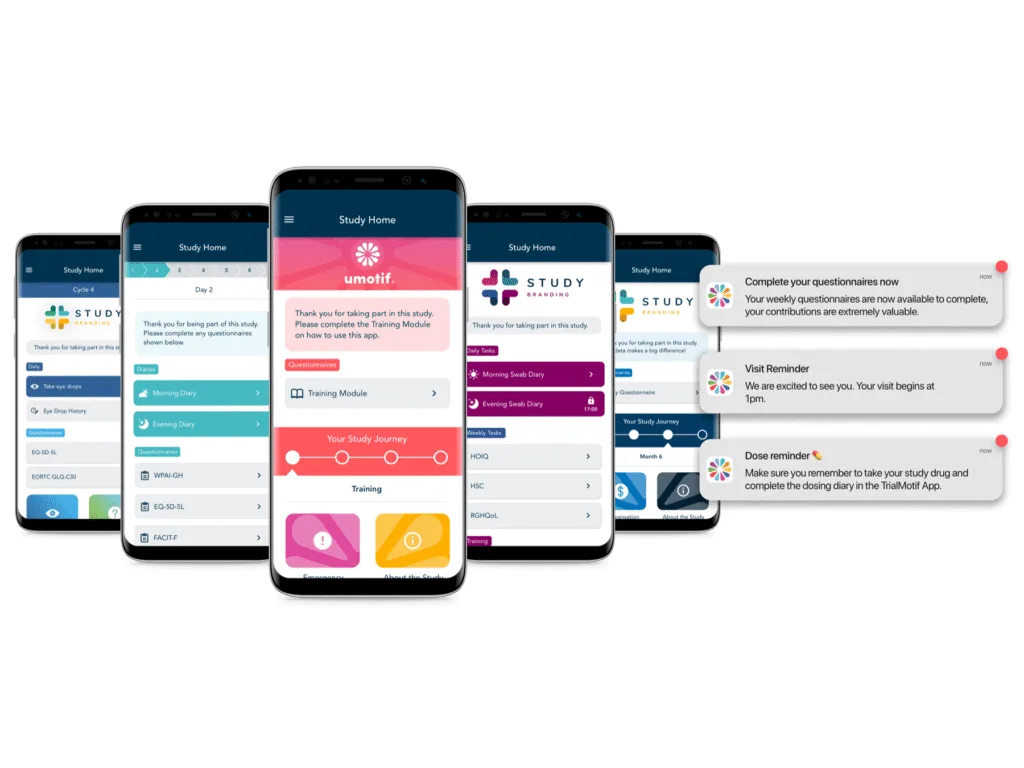
User-centric design for engaged participants
uMotif’s simple, intuitive, and accessible interface allows participants (or their caregivers) and sites to integrate data capture seamlessly into their daily lives.
Our design approach isn’t just about what the user sees, but what happens behind the scenes. Support implementation of even the most complex protocols with uMotif’s flexible, adaptive scheduling and dynamic alerting to minimize user burden.
Unrivaled Compliance
uMotif’s patient-centric design combines with flexible alerts and reminders to reduce missing data and deliver high compliance.
Our eCOA solution regularly achieves >90% compliance in both clinical and real-world studies, helping sponsors and CROs run faster, more efficient studies.
Major Contender in eCOA Products
uMotif was recognized in the in the 2025 Everest Group Life Sciences eCOA Products PEAK Matrix® Assessment and named a Major Contender.
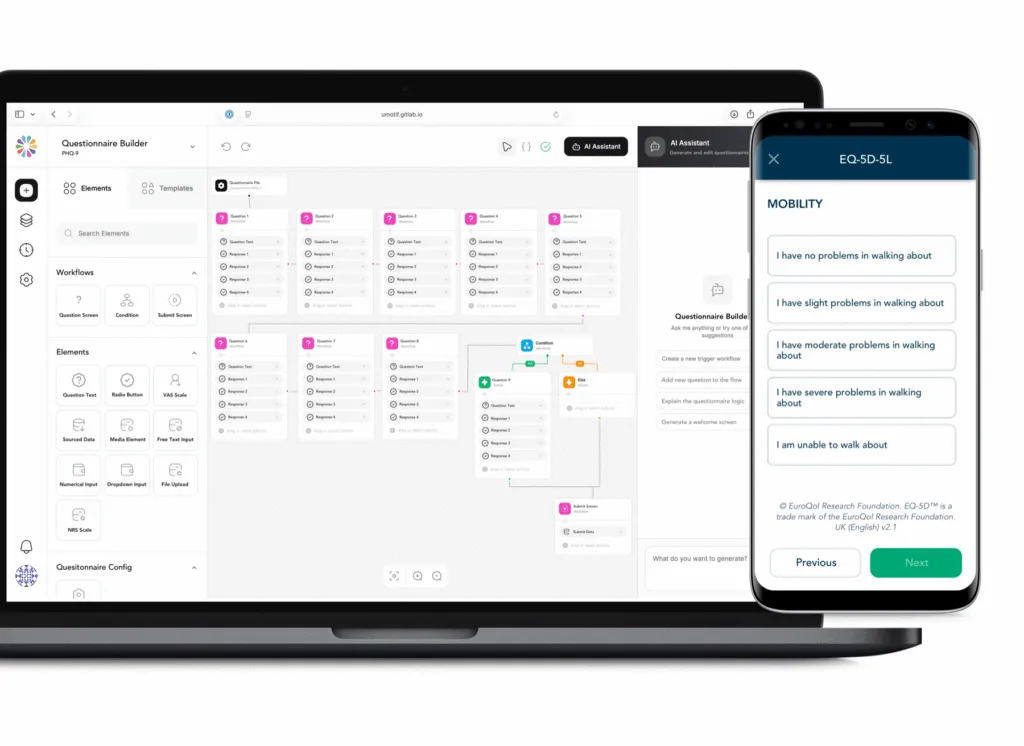
Enhanced questionnaire builder tools
Our eCOA questionnaire builder enables quick configuration of existing eCOAs, as well as bespoke questionnaires and home-grown eDiaries, with a built-in visual screen preview providing early visibility into screen design.
Linked to our library infrastructure, sponsor-specific repositories offer faster study build and deployment, designed to rapidly take studies from protocol to FPI.
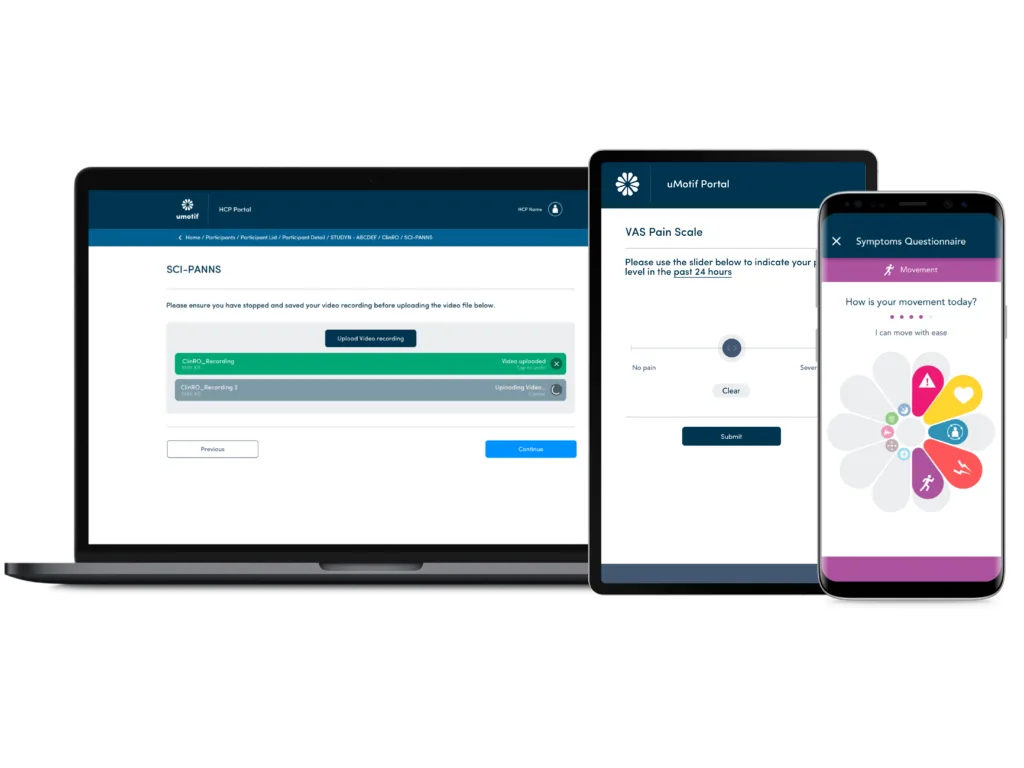
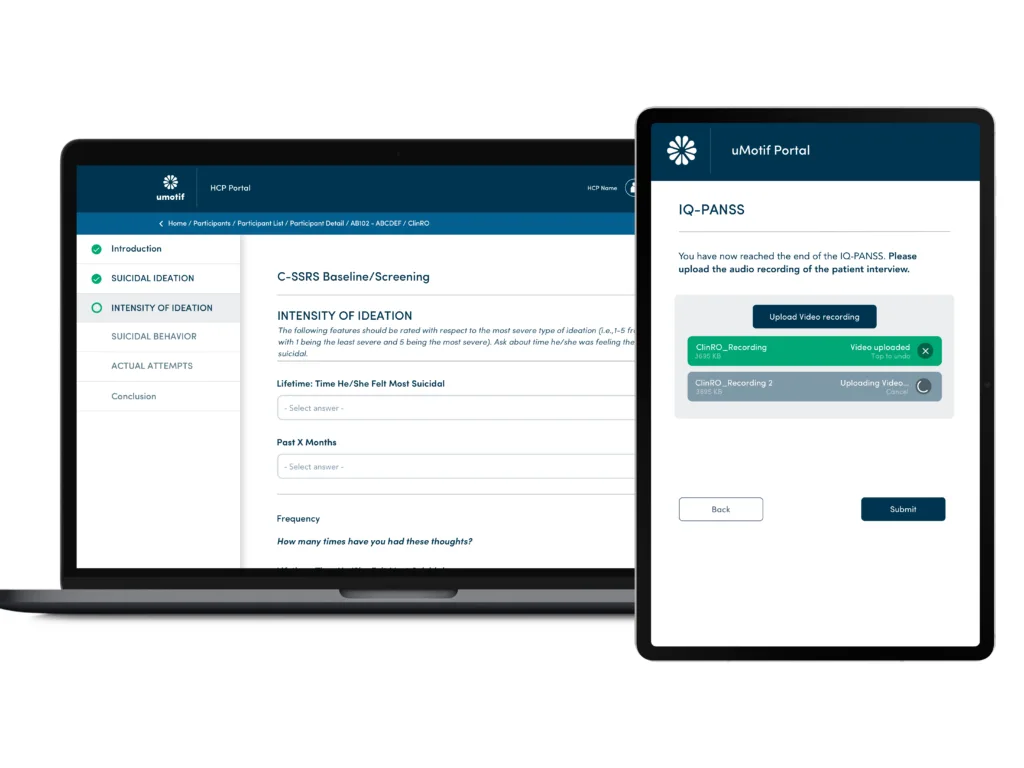
Complex ClinROs and Audio Upload
uMotif provides a robust, comprehensive system that takes a standardized approach to eClinRO administration while streamlining data capture for the rater. Role-based access control ensures the appropriate controls are in place so that the right raters have access to the right measures.
The uMotif system also supports audio upload of eClinRO administrations for quality review.
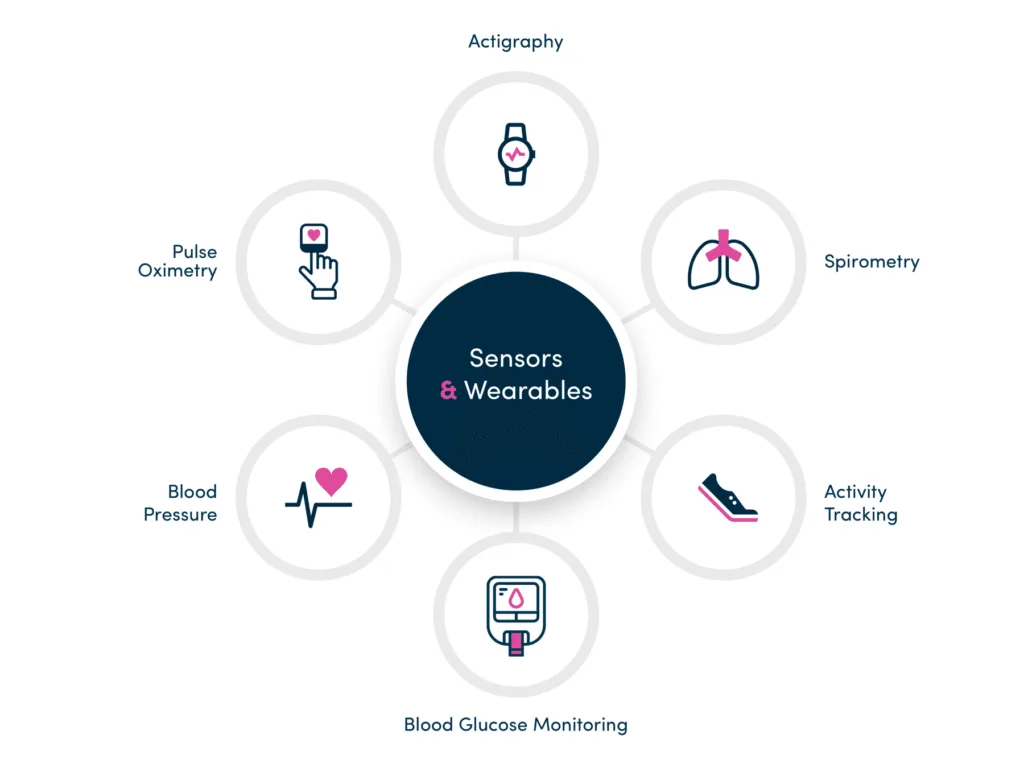
Sensors and Wearables
An extensible solution to quickly integrate sensors and wearables to capture more objective, real-time insights into participant functioning to support trial endpoints. Combining this data with electronic patient-reported outcomes (ePRO) offers a powerful picture of participant health.
The uMotif app can also house important training and support content/material on the set-up, use, and trouble-shooting of integrated digital health technologies (DHTs), ensuring help is always at hand.
Real-time Data Insights
Real-time recruitment, eligibility, eCOA compliance and device reporting through a powerful web-based portal to make study management and oversight simpler, faster and easier.


Underpinned by Scientific Excellence
Everything we do at uMotif is informed by Science. Find out more about our specific scientific services below:
- Scientific Consulting
- Scale Management and Translations
Additional solutions in the uMotif portfolio
One platform, stronger participation and stronger trials.
- eConsent and Telehealth — Flexible solutions for informed onboarding, consent and telehealth, to enhance the participant experience.
- Patient Engagement — Engagement solutions deployed alongside eCOA or standalone — supporting all phases of research.
- Patient Ride/Uber Health — To alleviate transportation challenges as a reason to not enroll in a clinical trial.
- Trial Awareness — Building trial visibility among HCP’s with potentially eligible patients.
- Integrations — Quickly integrate with key 3rd party systems, such as randomization solutions (IVRS, RTSM), EDC, and tokenization on a study-by-study basis.
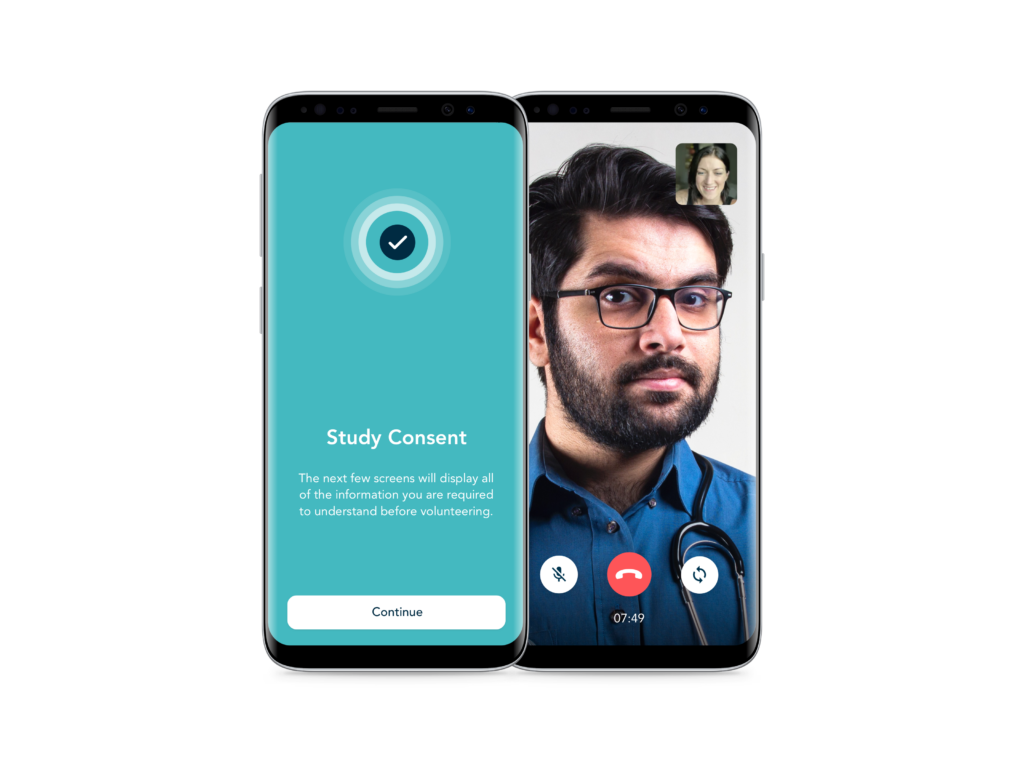
Validated eCOA / ePRO Clinical Data Capture

Provisioned, BYOD or Hybrid Device Approaches

24/7 Multi-Lingual Helpdesk

GCP and 21 CFR Part 11 Compliant

Sensors and Data Tokenization

Mobile, Tablet and Web Data Capture
All Languages Supported

IRT, EDC and Data Lake Integrations
