
An Introduction to Clinical Outcome Assessments (COAs)
In clinical research, a fundamental question is “How do we show whether a medical product works and is safe?” Clinical outcome assessments, commonly referred to as COAs, help answer this question.
A clinical outcome assessment is any measure that reflects or describes how a patient feels, functions or survives. Depending on the study, COAs may also be called:
- Questionnaires
- Surveys
- Instruments
- Scales
- Diaries
No matter the name, COAs are foundational tools in clinical trials.
Why Clinical Outcome Assessments Matter in Clinical Trials
COAs influence nearly every stage of a study — from determining who can participate to judging the safety and effectiveness of a treatment.
Eligibility Criteria
COAs may be part of inclusion or exclusion criteria, ensuring that participants are suitable for the study.
Efficacy Endpoints
COAs often serve as primary, secondary or exploratory endpoints that determine whether an intervention has had the intended effect.
Safety Endpoints and Monitoring
COAs often serve as safety endpoints in trials, and can also be used throughout the study to evaluate safety signals and monitor whether it is safe for a participant to continue in the trial.
Reimbursement Decisions
After a product reaches market, COA data can be used to support health-technology assessments and reimbursement decisions.
Clinical Outcome Assessment Examples
COAs are grouped into four major categories, each designed to capture a different perspective of a patient’s experience.
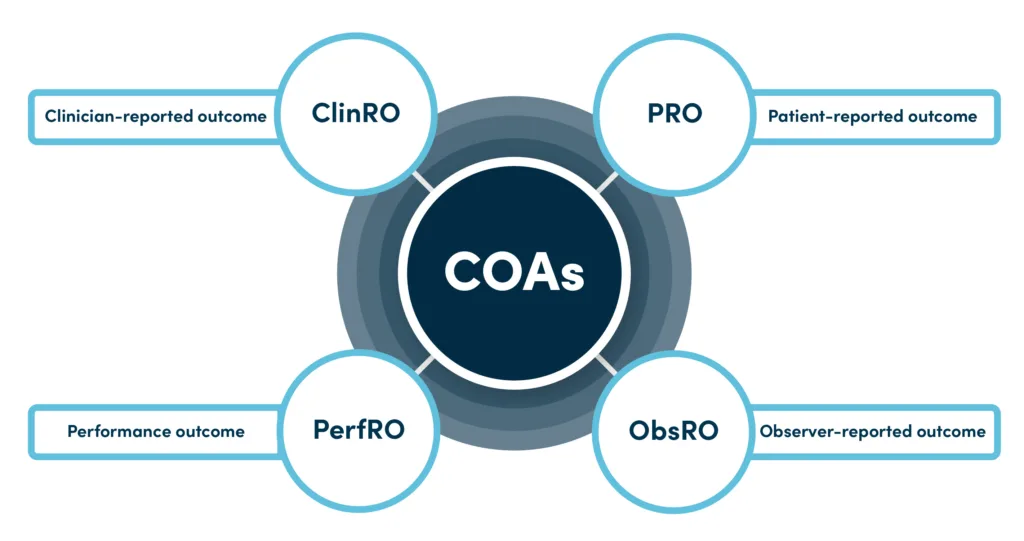
Clinician-Reported Outcomes (ClinRO)
A ClinRO is a measurement completed by a trained healthcare professional following direct observation, interview or physical examination of a participant.
Clinicians may evaluate:
- Physical symptoms
- Observable behaviors
- Disease manifestations
- Functional limitations
When Are ClinROs Appropriate?
ClinROs are ideal for conditions requiring expert assessment, such as:
- Skin diseases (e.g., psoriasis, eczema)
- Inflammatory joint diseases (e.g., rheumatoid or psoriatic arthritis)
- Neurological disorders (e.g., Alzheimer’s, Parkinson’s, multiple sclerosis)
They are also important when patients cannot reliably self-report due to:
- Cognitive impairment
- Developmental disabilities
- Psychosis (delusions, hallucinations)
- Very young age (infants/toddlers)
Common ClinRO Instruments
- Columbia-Suicide Severity Rating Scale (C-SSRS)
- Hamilton Depression Rating Scale (HAM-D)
- Eczema Area and Severity Index (EASI)
- Clinical Global Impressions:
- Severity (CGI-S)
- Change/Improvement (CGI-C/CGI-I)
- Schizophrenia (CGI-SCH)
Patient-Reported Outcomes (PRO)
A patient-reported outcome captures information directly from the patient, without interpretation from clinicians or caregivers, and are advised when measuring concepts best known by the patient themselves.
Today, PROs are routinely used as endpoints in clinical trials to elicit the patient perspective, ensuring greater patient focus on medical product development, and providing the opportunity to measure concepts that matter to the patient to really understand the value and treatment benefit (or risk) of a product.
Patients may complete PROs independently or via interview (as long as responses are recorded verbatim, and the interviewer does not add their own interpretation).
What Do PROs Measure?
PROs often assess:
- Symptoms
- Health-related quality of life
- Functional status
- Mood
- Treatment satisfaction

Common PRO Instruments Quality of Life Measures
- EORTC QLQ-C30
- EQ-5D-5L
- SF-36
Event-Driven or Daily Reporting
- Migraine diaries
- Seizure diaries
- Pain diaries
- Asthma diaries
- Allergy symptom diaries
When Should PROs Be Used?
Use PROs when:
- Patient perspective is essential
- Symptoms cannot be observed externally (e.g., pain, itch, nausea)
- Frequent measurement is required (daily/weekly)
- Quality of life or satisfaction needs evaluation
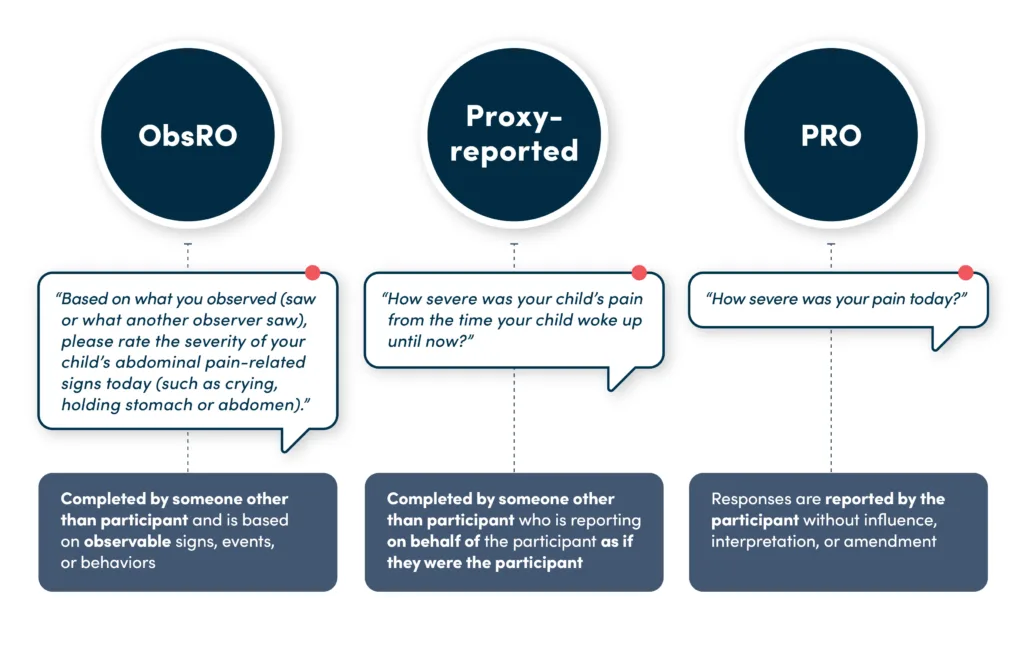
Observer-Reported Outcomes (ObsRO)
An ObsRO is reported by someone who regularly observes the patient — typically a parent or caregiver. It focuses only on observable behaviors or events, without medical judgment or interpretation.
ObsROs are particularly valuable for:
- Infants and young children
- Individuals with cognitive impairment
- Caregivers impacted by a participant’s condition
ObsRO Examples
Quality of Life
- PedsQL (Pediatric Quality of Life Inventory)
Event Reporting
- Seizure diaries
- Bleed diaries
Behavioral Measures
- BASC (Behavior Assessment System for Children – Parent Report)
- Alzheimer’s Disease Cooperative Study – Activities of Daily Living
ObsROs are distinct from proxy-reported outcomes. Proxy-reported outcome measures involve someone other than the patient reporting on the patient experience as if they were the patient. These are strongly discouraged by regulatory bodies, because they are measuring concepts only known to the patients themselves, and are not necessarily a true reflection of how the patient feels or functions.
Concepts that are only known by the patient (e.g., pain, symptoms, feelings), should be measured by a PRO, or observable behavior by an ObsRO.
What Is a Proxy-Reported Outcome?
Performance-Reported Outcomes (PerfO)
A PerfO is based on a standardized task that a patient performs, typically under structured conditions.
May be administered by an appropriately trained individual or completed by patient independently.
These tasks assess:
- Physical capabilities
- Cognitive function
- Sensory/perceptual processing
PerfOs may require equipment such as:
- Neuropsychological testing kits
- Walking courses
- Stopwatches
- Digital sensors
Common PerfO Measures Cognitive Tests
- Alzheimer’s Disease Assessment Scale-Cognitive (ADAS-Cog)
- Montreal Cognitive Assessment (MoCA)
- Mini Mental State Exam (MMSE)
- Symbol Digit Modalities Test (SDMT)
Physical Performance
- 25-Foot Walk Test
- 6-Minute Walk Test
The Value of Clinical Outcome Assessments
Clinical outcome assessments are crucial tools that bring patient experience, clinical expertise and measurable performance together. Whether reported by clinicians, patients, caregivers or demonstrated through performance tests, COAs provide rich insights into the real impact of medical products.
By using the right COA at the right time, clinical trials can more accurately capture what matters most to patients. Electronic data capture is a robust way to capture COA data — find out more about the benefits here.