eCOA Supporting Parkinson’s Research
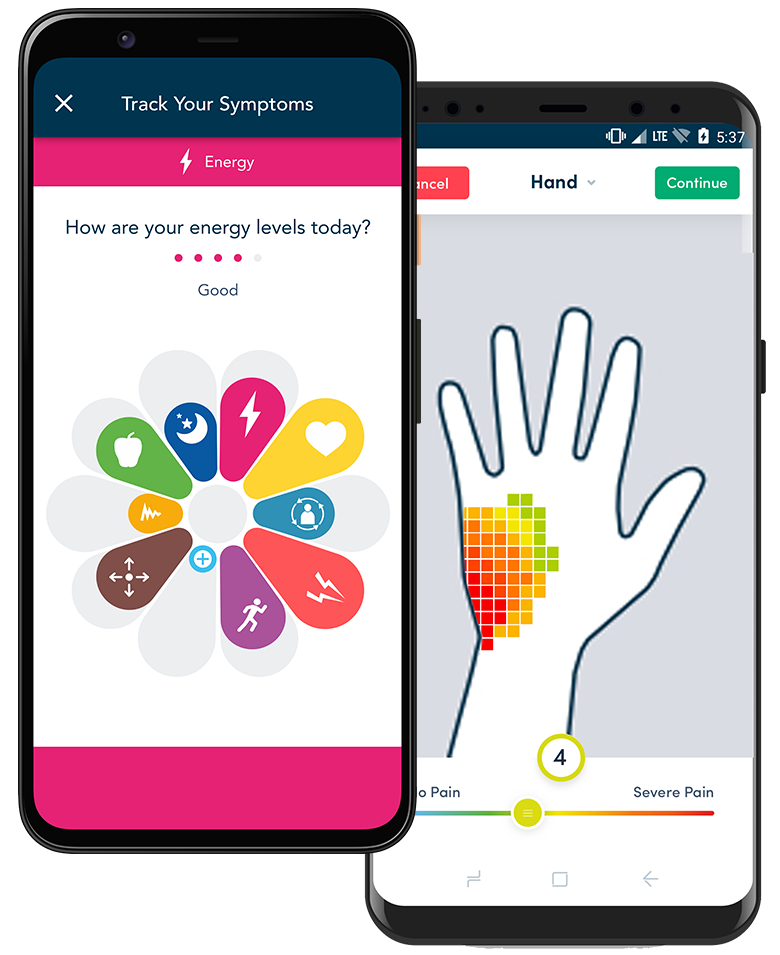
An unmet need for Parkinson’s patients, who wanted help in tracking their daily symptoms, became the foundation for uMotif’s patient-centric eCOA/ePRO and engagement platform.

Our unparalleled understanding of the burden that trial participation puts on Parkinson’s patients enabled us to develop eCOA/ePRO technology that yields high compliance and quality data.

Case Study: 100 for Parkinson’s
Learn how uMotif’s eCOA/ePRO platform powered a multi-nation decentralized study, helping people with Parkinson’s Disease better understand symptoms, and creating a strong data foundation for future research.
This large scale study engaged more than 4,200 patients, across 60 countries, capturing more than 2.2 million data points and tracking over 1.3 million symptom scores.
Cathy’s story: eCOA supporting Parkinson’s research
Hear the story of Cathy, a Parkinson’s patient, and how using the uMotif app not only enabled her to take part in this study but is having a positive impact on the management of her condition, including:
- The ability to track her symptoms
- The use of the app to help her clinician understand her day-to-day symptoms
- Assisting her healthcare providers to optimize her medication
View a demo
Sign up now to view a Demo of the uMotif eCOA/ePRO platform, and see how our unique, patient centered design works