
QUASAR case study
 Powering the largest remote study investigating sleep and quality of life for adults with rheumatoid arthritis.
Powering the largest remote study investigating sleep and quality of life for adults with rheumatoid arthritis.
“We are seeing unprecedented volumes of data. The entire team is astounded by the numbers and impressed by the uMotif platform.”
Dr Katie Druce Centre for Epidemiology, University of Manchester
 Background
Background

The University of Manchester conducted a real-world evidence (RWE) prospective cohort study designed to understand the burden of sleep disruption on rheumatoid arthritis sufferers.
The study, named QUASAR, required a modern technology platform to accurately capture data via an easy-to-use mobile app downloaded on patients’ own devices.
University of Manchester’s approach included:
– Implement a hybrid site and virtual-based trial
– Embed data collection into patients’ daily lives via mobile technology
– Capture a minimum of two validated questionnaires, 15 days of symptom data, and one sleep diary
– Supplement subjective data capture with a sleep tracking device
 The Challenge
The Challenge

– Perform the largest remote study into sleep and rheumatoid arthritis
– Avoid using sleep clinics
– Provide an engaging patient experience
– Capture population representative data
 Impact
Impact

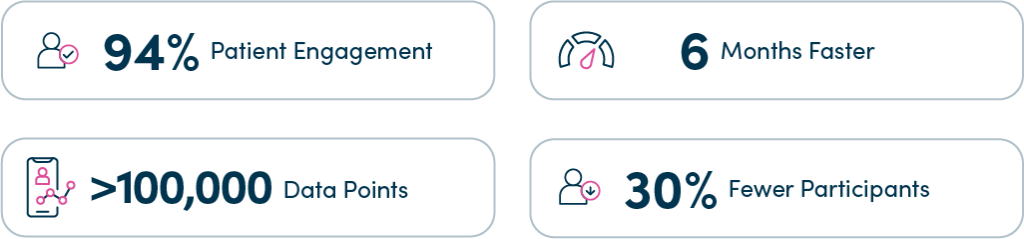
Implementing uMotif’s platform enabled the QUASAR research team to complete the study six months early and required 30% fewer participants than expected due to high levels of engagement and complete data submission.
Capturing high volumes of quality data
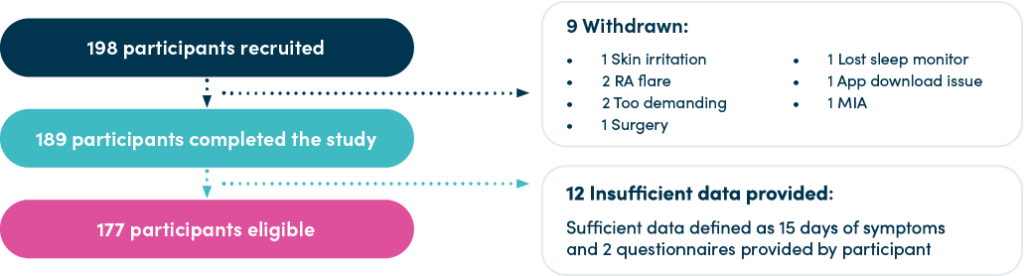
Using a hybrid model encompassing on-site and virtual data collection, a group of 177 patients (94% of eligible participants) submitted complete data providing more than 100,000 data points. The high volume and quality of data captured is unprecedented in similar studies.
Putting patients at the center
In using a data capture mobile app, patients contributed to clinical research from the comfort of their own homes. This provided more realistic and accurate sleep data, while removing the need and expense of using sleep clinics.
Patients loved using the mobile app, finding it easy and fun to use. In-app data visualizations of symptoms, as well as a sleep diary, provided participants with new insights into their condition without biasing the study.
Minimum study power reached early
Higher patient engagement levels enabled the research team to recruit fewer patients to achieve the data volume required for powering the study. In total, 198 participants were recruited (compared to the 350 patients that the protocol initially expected).
New data insights
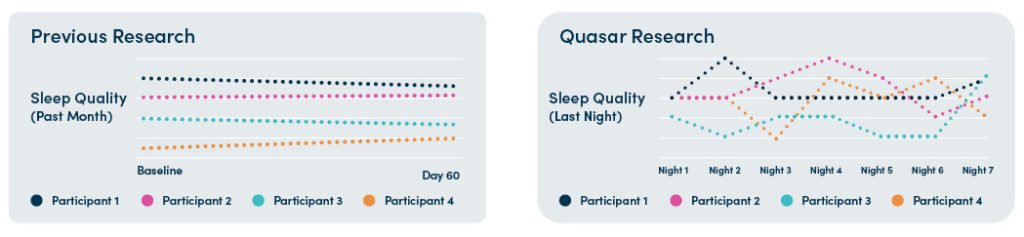
Previous research has only highlighted macro trends of sleep quality over time. In collecting more data, with greater accuracy, researchers are now able to identify deeper patterns in the relationship between rheumatoid arthritis symptoms and sleep.
Find out more about how uMotif can help drive unparalleled patient engagement and data capture in your next study.